What is Benlysta (belimumab) for?
Benlysta (belimumab) is a treatment for:
- Children aged 5 years and older with active, autoantibody-positive, systemic lupus erythematosus (SLE; lupus)
- Adult patients with active lupus nephritis (LN)
It is used with other treatments to help control the immune system.
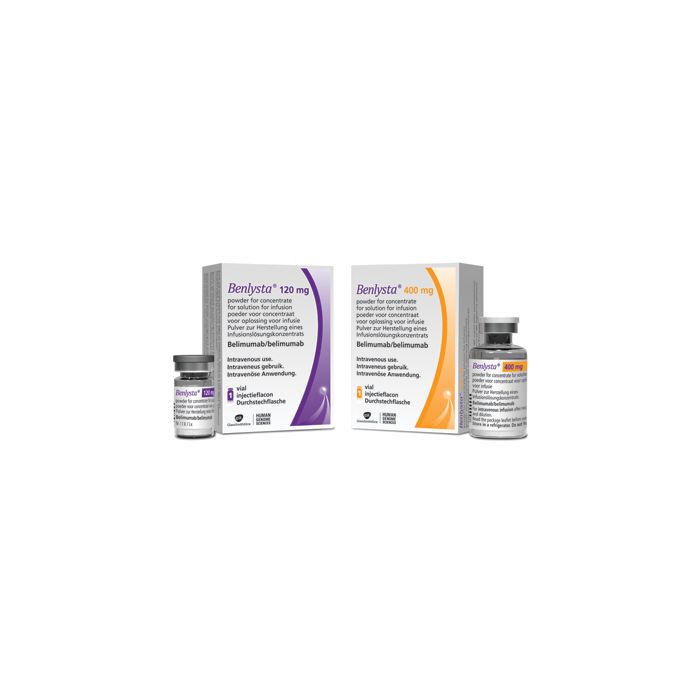
It comes in two forms: a 120 mg vial for IV use and a 400 mg vial for IV use. It also comes as a 200 mg pen and syringe for injection under the skin.
How does Benlysta (belimumab) work?
About 50% of people with SLE get lupus nephritis (LN). LN damages the kidneys and raises the risk of kidney failure, heart problems, and death.
B cells are key in SLE inflammation. They make antibodies to fight infections but also attack the body’s cells and organs in lupus.
Belimumab blocks BLyS, a protein that helps B cells live. By doing this, it stops autoreactive B cells from surviving.
Where has Benlysta (belimumab) been approved?
Benlysta (belimumab) is approved as an add-on therapy by:
- The Food and Drug Administration (FDA), USA:
- On 9 March 2011 as intravenous (IV) formulation for adults with lupus.
- On July 21, 2017 as subcutaneous (SC) formulation for adults with lupus.
- On April 26, 2019 as IV formulation for children aged ≥5 years with lupus.
- On December 17, 2020 as IV and SC formulation for adults with active LN.
- The European Medicines Agency (EMA), Europe:
- On May 19, 2011 as IV formulation for adults with lupus.
- On November 13, 2017 as SC formulation for adults with lupus.
- On September 20, 2019 as SC formulation for children aged ≥5 years with lupus.
The FDA approved Benlysta under Priority Review for lupus. It also got Breakthrough Therapy Designation and Priority Review for LN treatment.
This medicine might be approved in other places too. If you have questions, contact our support team.
How is Benlysta (belimumab) taken?
Benlysta (belimumab) can be taken in two ways:
- Via subcutaneous self-injection at home for adults with SLE and/or LN:
- A standard dose of 200 mg given once weekly
- Intravenous infusion given by a healthcare provider for adults with SLE and/or LN and for children (aged ≥5 years) with SLE.
- A standard dosage regimen is 10 mg/kg every 2 weeks for the 3 three doses. IV treatment is given over approximately one hour.
- After this, this dose is given every 4 weeks
It is used with other treatments.
Talk to your doctor about the best option for you. Adults can switch from IV to at-home treatment.
For full details on Benlysta (belimumab) dosage and how to use it, check the official prescribing information in our references section.
Note: Always talk to your doctor about the right dose for you.
Are there any known adverse reactions or side effects of Benlysta (belimumab)?
Common adverse reactions
The most common side effects ( ≥5% of patients) include:
- High temperature or fever (pyrexia)
- Low white blood cell count (can be seen in blood tests)
- Nose, throat or stomach infection
- Pain in hands or feet
- Migraine
- Depression
- Injection site reactions
Serious adverse reactions
The serious side effects listed include:
- Increased risk of infections, including a rare but serious brain infection called progressive multifocal leukoencephalopathy (PML)
Use in a specific population
It’s not clear if Benlysta (belimumab) is safe for a fetus. Women who might get pregnant should use birth control while taking it and for 4 months after stopping.
For a full list of side effects and adverse reactions, see the official prescribing information.






Reviews
There are no reviews yet.