What is Cabenuva (cabotegravir + rilpivirine) for?
Buy Cabenuva (cabotegravir + rilpivirine). Cabenuva (cabotegravir + rilpivirine) is a treatment for HIV-1 infection. It’s for people who need a new HIV treatment. Their doctor must check if they meet certain requirements first.
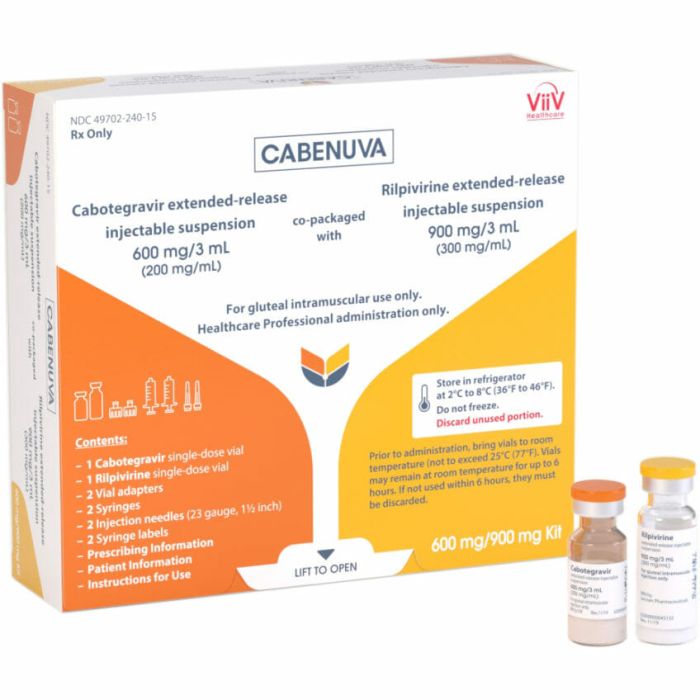
It comes in a vial of cabotegravir suspension and a vial of rilpivirine suspension. The options are 400/600mg or 600/900mg.
How does Cabenuva (cabotegravir + rilpivirine) work?
Cabotegravir, in Cabenuva, blocks an enzyme called integrase. This enzyme is used by the virus to replicate. By blocking integrase, cabotegravir reduces HIV particles in the blood.
Rilpivirine, the other active ingredient, blocks reverse transcriptase (RT). RT is an enzyme the virus uses to make new copies. Rilpivirine lowers HIV particles by blocking RT.
This medicine can’t cure HIV or AIDS. But it helps the immune system repair itself and prevents further damage.
Where has Cabenuva (cabotegravir + rilpivirine) been approved?
Cabenuva was approved for HIV-1 treatment by:
- Health Canada on March 18, 2020
- The Food and Drug Administration (FDA), USA on January 21, 2021.
- Therapeutic Goods Administration (TGA), Australia on February 23, 2021.
This medicine might be approved in other places too. If you have questions, contact our support team.
How is Cabenuva (cabotegravir + rilpivirine) taken?
The dosage is as follows:
- First, a one-month starter pill regimen is given. This includes one 30-mg tablet of Vocabria and one 25-mg tablet of Edurant.
- On the last day of Month 1, patients get their first injections. They receive 600 mg of cabotegravir and 900 mg of rilpivirine.
- Starting in Month 3, patients get monthly injections. They receive 400 mg of cabotegravir and 600 mg of rilpivirine.
The injections are given in the buttocks by a healthcare professional. They are given one in each side.
Cabenuva may interact with other medicines. It’s important to tell your doctor about all medicines you take. The medicine stays in your blood for up to 12 months or longer.
For more details on dosage and administration, see the official prescribing information. Note: Always consult your doctor for personalized dosing.
Are there any known adverse reactions or side effects of Cabenuva (cabotegravir + rilpivirine)?
Common adverse reactions
The most common side effects ( ≥2% of patients) listed in the prescribing information include:
- Injection site reactions
- Fever (pyrexia)
- Tiredness (fatigue)
- Headache
- Muscle pain
- Nausea
- Sleep disorders
- Dizziness
- Rash
Serious adverse reactions
The serious adverse reactions listed in the prescribing information include:
- Serious injection site reactions
- Liver problems
- Depression or mood changes
Use in a specific population
It is not known if Cabenuva (cabotegravir + rilpivirine) can harm a fetus. The medicine may stay in your blood for up to 12 months or longer.
Mothers with a HIV-1 infection should not breastfeed. The virus can pass to the baby through breast milk.
For a comprehensive list of side effects and adverse reactions, please refer to the official prescribing information.