What is Dupixent (dupilumab) for?
Buy Dupixent (dupilumab). Dupixent (dupilumab) is a treatment for several conditions. It works by blocking IL-4 and IL-13 proteins. These proteins cause inflammation in the skin and airways.
- It treats atopic dermatitis (eczema) in people aged 6 and up. It’s for those who don’t get better with other treatments.
- It’s also for asthma in people aged 12 and up. This includes those who need steroids to control their asthma.
- Adults with chronic sinusitis and nasal polyps can use it too. It’s for those who don’t get better with other treatments.
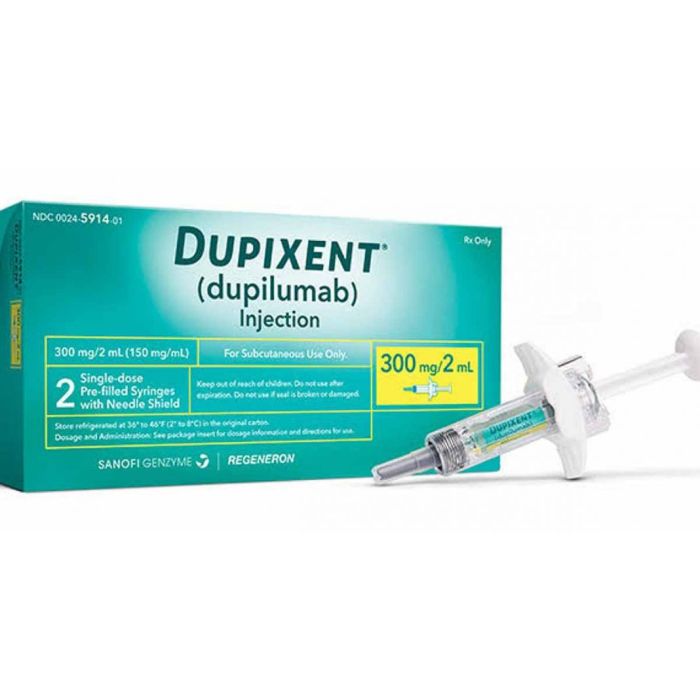
It comes in a syringe or pen. You can give yourself a shot under the skin. The syringe has 300 mg or 200 mg, and the pen has 300 mg.
How does Dupixent (dupilumab) work?
Diseases like atopic dermatitis, asthma, and sinusitis have inflammation at their core. High levels of IL-4 and IL-13 proteins cause this inflammation.
Dupixent’s active ingredient, dupilumab, is a protein that blocks these proteins. By doing this, it stops IL-4 and IL-13 from causing inflammation. This relieves symptoms of the disease.
Where has Dupixent (dupilumab) been approved?
Dupixent has been approved in several countries. It’s for treating atopic dermatitis, asthma, and sinusitis.
- The FDA approved it in the USA on:
- March 28, 2017 for atopic dermatitis.
- October 19, 2018 for asthma.
- June 26, 2019 for sinusitis.
- May 26, 2020 for children with atopic dermatitis.
- The EMA approved it in Europe on:
- September 28, 2017 for atopic dermatitis.
- May 7, 2019 for asthma.
- October 29, 2019 for sinusitis.
- November 30, 2020 for children with atopic dermatitis.
The FDA gave it Priority Review and Breakthrough Therapy status. This status helps speed up its development and review.
The FDA also approved it for eosinophilic esophagitis in people aged 12 and up. It’s currently being reviewed for asthma in children aged 6 to 11.
This medicine might be approved in other places too. If you have questions, contact our support team.
How is Dupixent (dupilumab) taken?
Atopic Dermatitis
The usual dose is:
- An initial dose of 600 mg (two 300 mg injections in different spots),
- Then 300 mg every 2 weeks.
For those under 60 kg, the dose or frequency might change.
Asthma
The standard dosage is:
- An initial dose of 400 mg (two 200 mg injections in different injection sites), followed by 200 mg given every 2 weeks, or
- An initial dose of 600 mg (two 300 mg injections in different injection sites), followed by 300 mg given every 2 weeks
Chronic Rhinosinusitis with Nasal Polyposis
The standard dosage is:
- 300 mg given every other week
Dupixent (dupilumab) is given by an injection under the skin (subcutaneous).
Patients may self-inject Dupixent, or a caregiver may give the injection. Before administering the medicine at home, it is important to receive proper training from a healthcare professional.
Complete information about Dupixent (dupilumab) dosage and administration can be found in the official prescribing information listed in our references section.
Note: Please consult with your treating doctor for personalised dosing.
Are there any known adverse reactions or side effects of Dupixent (dupilumab)?
Common adverse reactions
The most common side effects ( ≥1% of patients) listed in the prescribing information include:
- Injection site reactions
- Conjunctivitis
- Blepharitis
- Oral herpes
- Keratitis
- Eye pruritus
- Other herpes simplex virus infection
- Dry eye
Serious adverse reactions
The serious adverse reactions listed in the prescribing information include:
- Allergic reactions
- Eye problems
Use in a specific population
Available data from use in pregnant women have not identified a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Human IgG antibodies are known to cross the placental barrier; therefore, this drug may be transmitted from the mother to the developing fetus.
For a comprehensive list of side effects and adverse reactions please refer to the official prescribing information.