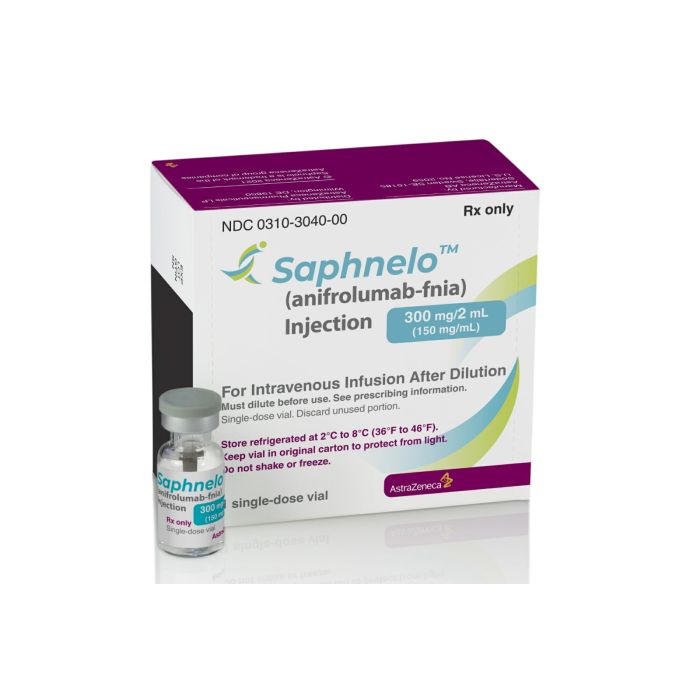
What is Saphnelo (anifrolumab) for?
Buy Saphnelo (anifrolumab). Saphnelo (anifrolumab) is a new treatment for systemic lupus erythematosus (SLE). It helps those with moderate to severe SLE who are already on standard treatments.
It comes in a vial form, with 300 mg/2 mL (150 mg/mL) anifrolumab in each.
How does Saphnelo (anifrolumab) work?
SLE is an autoimmune disease, the most common form of lupus. In SLE patients, the immune system makes antibodies that can harm any part of the body. This can damage multiple organs.
Anifrolumab blocks type I interferons (IFN), proteins made by the immune system. These proteins are a main cause of tissue damage and disease progression. Most SLE patients have high levels of type I IFN, which worsens symptoms and disease severity.
Where has Saphnelo (anifrolumab) been approved?
Saphnelo (anifrolumab) has been approved for SLE treatment by:
The Food and Drug Administration (FDA), USA on August 2, 2021.
It was approved under Priority Review and was granted Fast Track designation by the FDA in 2016.
It’s also under review in the EU and Japan for SLE. A Phase 3 trial using subcutaneous delivery has started. More Phase 3 trials are planned for lupus nephritis, cutaneous lupus erythematosus, and myositis.
Please note that this medicine may have also been approved in other regions than the ones we’ve listed. If you have a question about its approval in a specific country feel free to contact our support team.
How is Saphnelo (anifrolumab) taken?
The standard dosage is:
300 mg given through a needle placed in a vein (IV or intravenous infusion) once every 4 weeks.
It takes about 30 minutes to administer the full dose.
Avoid the use of live or live-attenuated vaccines during treatment with Saphnelo (anifrolumab).
Saphnelo (anifrolumab) is not recommended for use in combination with other biologic therapies, including B-cell targeted therapies.
Complete information about Saphnelo (anifrolumab) dosage and administration can be found in the official prescribing information listed in our references section.
Note: Please consult with your treating doctor for personalised dosing.
Are there any known adverse reactions or side effects of Saphnelo (anifrolumab)?
Common adverse reactions
The most common side effects (≥5% of patients) listed in the prescribing information include:
- upper respiratory infections
- infusion reactions
- cough
- bronchitis
- shingles (herpes zoster)
Serious adverse reactions
The serious adverse reactions listed in the prescribing information include:
- serious infections
- allergic (hypersensitivity) reactions
- cancer
Use in a specific population
It is not (yet) known if Saphnelo (anifrolumab) can cause major birth defects or miscarriage. Females who are able to become pregnant are advised to inform their treating doctor if they intend to become pregnant during therapy, suspect they are pregnant or become pregnant while receiving this medicine.
The safety and efficacy of Saphnelo (anifrolumab) in patients younger than 18 years of age has not been established.
For a comprehensive list of side effects and adverse reactions please refer to the official prescribing information.