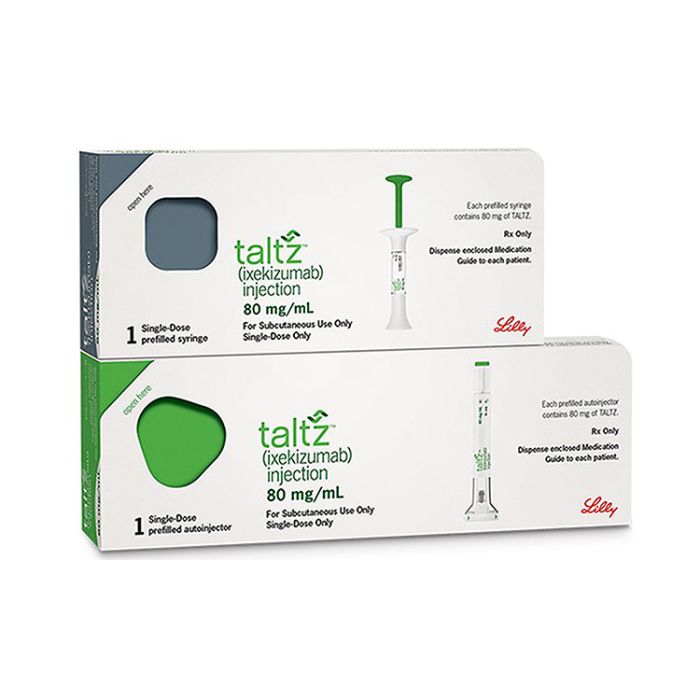
What is Taltz (ixekizumab) for?
Buy Taltz (ixekizumab). Taltz (ixekizumab) is a treatment for:
- moderate to severe plaque psoriasis for those ready for systemic therapy or phototherapy
- Active psoriatic arthritis in adults who didn’t get better with other treatments.
How does Taltz (ixekizumab) work?
Taltz’s main ingredient, ixekizumab, is a protein that blocks interleukin 17A. This is a key messenger in the immune system.
Interleukin 17A causes inflammation, leading to psoriasis and psoriatic arthritis. By blocking it, ixekizumab reduces the immune system’s activity. This helps lessen the symptoms of these conditions.
Where has Taltz (ixekizumab) been approved?
Taltz (ixekizumab) has been approved by:
- Food and Drug Administration (FDA), USA:
- March 22, 2016, for plaque psoriasis
- December 1, 2017, for psoriatic arthritis
- European Medical Agency (EMA), European Union:
- May 2, 2016, for plaque psoriasis
- January 11, 2018, for psoriatic arthritis
- Therapeutic Goods Administration (TGA), Australia, September 6, 2016, for plaque psoriasis
- Health Canada, August 11, 2016, for plaque psoriasis
This medicine might also be approved in other places. If you have questions, contact our support team.
How is Taltz (ixekizumab) taken?
For plaque psoriasis, the dosage is:
- first dose of 160 mg (two subcutaneous injections)
- for the first 12 weeks: a subcutaneous injection of 80 mg every 2 weeks
- thereafter a subcutaneous injection of 80 mg every 4 weeks.
For psoriatic arthritis, the dosage is:
- first dose of 160 mg (two subcutaneous injections)
- thereafter a subcutaneous injection of 80 mg every 4 weeks.
Taltz (ixekizumab) comes as a prefilled syringe or pen. Instructions for use are in our resources section.
For full details on dosage and administration, see the official prescribing information in our references section.
Always talk to your doctor for personalized advice on dosage.
Are there any known side effects of Taltz (ixekizumab)?
Common side effects
The most common side effects include:
- injection site reactions
- upper respiratory tract infections
- nausea (feeling sick)
- tinea infections (superficial fungal infection of the skin)
Serious side effects
The serious side effects listed in the prescribing information include:
- serious infections
- hypersensitivity (serious allergic reaction)
- inflammatory Bowel Disease
Use in a specific population
Taltz (ixekizumab) must not be given to patients with active tuberculosis (TB). Anti-TB therapy should be considered in patients with latent TB prior to initiation of treatment.
There is a limited knowledge about the use of ixekizumab in pregnant women. As a precautionary measure, it is preferable to avoid the use of Taltz during pregnancy. Use of effective contraception during treatment and for at least 10 weeks after treatment is recommended.
Breast-feeding should be avoided during treatment.