What is Givlaari (givosiran) for?
Givlaari (givosiran) is a treatment for adults and kids over 12 with acute hepatic porphyria (AHP). It’s a special RNA that helps manage this rare genetic disorder.
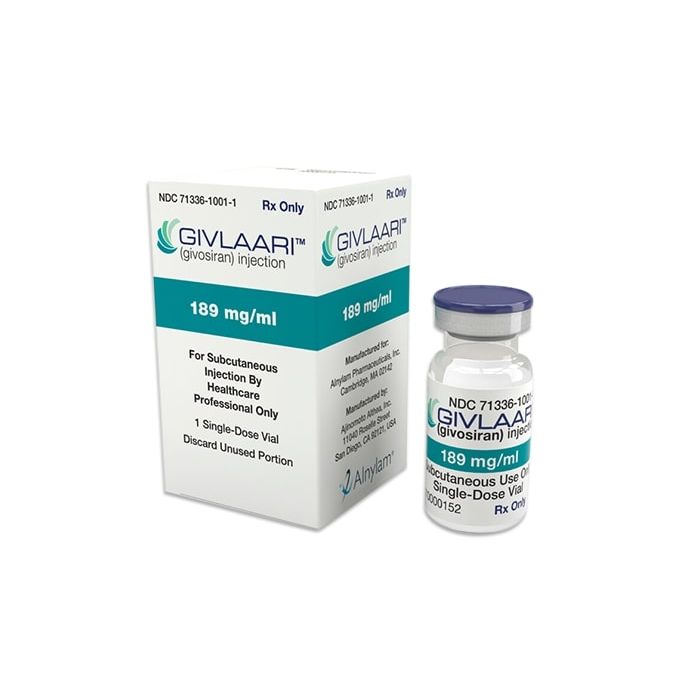
It comes in a single-dose vial with 189 mg/ml givosiran.
How does Givlaari (givosiran) work?
AHP is a rare genetic disorder where the heme pathway doesn’t work right. Heme is key for carrying oxygen in the blood. The liver controls this pathway with the enzyme aminolevulinate synthase 1 (ALAS1).
Triggers can make ALAS1 levels go up. This leads to harmful molecules like aminolevulinic acid (ALA) and porphobilinogen (PBG). These molecules hurt nerve cells, causing AHP symptoms and attacks.
Givosiran is a special RNA that lowers ALA and PBG levels. This protects patients from AHP attacks and symptoms.
Where has Givlaari (givosiran) been approved?
Givlaari (givosiran) has been approved by:
- The Food and Drug Administration (FDA), USA, on November 20, 2019 for adults with AHP.
- The European Medicines Agency (EMA), on January 31, 2020 for adults and kids over 12 with AHP.
- The Pharmaceuticals and Medical Devices Agency (PMDA), Japan on June 23, 2021.
The FDA gave it Priority Review and Breakthrough Therapy designation. It also got Orphan Drug designation from the FDA and EMA to help rare disease treatments.
This medicine might be approved in other places too. If you have questions, contact our support team.
How is Givlaari (givosiran) taken?
The usual dose is:
- 2.5 mg/kg actual body weight given via subcutaneous injection once a month.
If you miss a dose, take it as soon as you can. Then, start monthly dosing again.
If liver enzyme levels go up a lot, take 1.25 mg/kg once a month. If levels go back down, you can go back to 2.5 mg/kg once a month.
For more details on dosage and how to take it, see the official prescribing information in our references section.
Note: Always talk to your doctor about the right dose for you.
Are there any known adverse reactions or side effects of Givlaari?
Common adverse reactions
The most common side effects (≥20% of patients) include:
- Nausea
- Injection site reactions
Serious adverse reactions
The serious side effects listed include:
- Severe allergic reactions (anaphylactic reactions)
- Liver problems
- Kidney problems
- Injection site reactions
Use in a specific population
Givlaari (givosiran) can harm a fetus if a pregnant woman takes it. It’s best to avoid pregnancy and breastfeeding.
For a full list of side effects and adverse reactions, see the official prescribing information.




Reviews
There are no reviews yet.