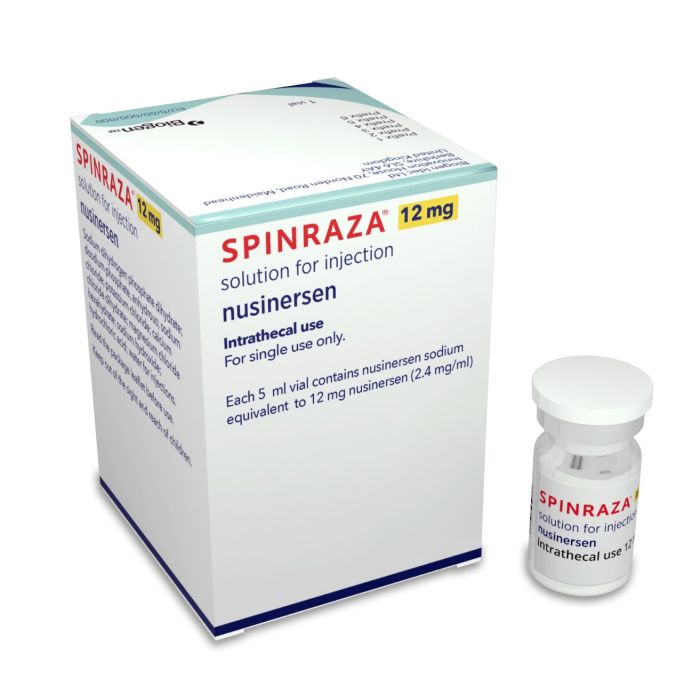
What is Spinraza (nusinersen) for?
Spinraza (nusinersen) is used to treat SMA in kids and adults.
How does Spinraza (nusinersen) work?
SMA is a rare disease that causes muscle weakness and wasting. It happens because motor neurons controlling movement are lost.
Spinraza (nusinersen) is a special medicine. It helps by making more of a protein called SMN. This protein is needed for motor neurons to work right.
Is Spinraza (nusinersen) approved?
Yes, Spinraza (nusinersen) is approved for SMA treatment. It was okay’d by:
- FDA (USA) on December 23, 2016
- EMA (EU) on May 30, 2017
- TGA (Australia) on November 2, 2017
- Health Canada on August 29, 2017
- Medsafe (New Zealand) on August 23, 2018
It’s also approved in China (February 2019). This list might not cover all places where it’s approved. For more info, contact our support team.
How do I take Spinraza (nusinersen)?
Spinraza is given as an injection into the spinal fluid. The dose is:
- 12 mg / 5 mL per shot
Start with 4 doses right away. Then, get doses every 2, 4, and 9 weeks. After that, get a dose every 4 months. Keep getting it as long as it helps.
For more details on how to take Spinraza (nusinersen), see the references section.
Always talk to your doctor about your specific needs.
Common side effects of Spinraza (nusinersen)
Some common side effects are:
- back pain
- headache
- vomiting.
What are the benefits of Spinraza (nusinersen)?
Spinraza has shown amazing results in clinical trials. Many patients have seen big improvements in their motor function and quality of life. Some patients have even learned to sit, stand, and walk on their own, which was once thought impossible.
Spinraza is also well-tolerated with few side effects. This makes it a promising treatment for those with SMA.
Is Spinraza (nusinersen) suitable for all SMA patients?
Spinraza is not right for every SMA patient. It’s crucial for those with SMA to talk to their healthcare provider about it. They need to consider the disease’s severity, age, and overall health.
What is the future of Spinraza (nusinersen)?
The future of Spinraza is promising. Researchers are working hard to make it even better and use it for more conditions. It’s already approved for SMA and is being tested for other neuromuscular disorders.
In conclusion, Spinraza has changed the game for SMA treatment. It offers hope and better lives for those with this disease. Its success shows the impact of medical innovation on rare genetic disorders.








Reviews
There are no reviews yet.